1.1 General Introduction
Numerous endogenous hormones are known to be secreted in a pulsatile manner. For
example, a neuroendocrine network controls reproduction and sexual development in
mammals, where the release of hormones is not constant with time. An important
reproductive hormone in human puberty and reproduction, gonadotropin releasing
hormone (GnRH), is released in rhythmic pulses from the hypothalamus every 1–2 hours
to stimulate secretion of luteinizing hormone (LH) and follicle stimulating hormone
(FSH) in the anterior pituitary gland. GnRH subsequently degrades within a few minutes
while the downstream signals carry out their actions in the human body. Disruption of
this rhythm is associated with pathologies in reproductive function and sexual
development. Central precocious puberty occurs when the GnRH pulse generator is
initiated too early in development. Conversely, a developmental delay of GnRH release
gives rise to delayed puberty [1]. Kallmann syndrome (KS) associated with anosmia, will
also present if GnRH secretion is reduced [2]. KS is diagnosed in about 1 in 30,000 males
and 1 in 120,000 females. Additionally, depressed GnRH secretion increases the risk of
osteoporosis in both man and woman. To mitigate the pathologies associated with GnRH
depression, several strategies involving GnRH release have been pursued [3-5]. Any such
strategy must restore not only appropriate levels of GnRH but also their pulsatility.
1.2 Background
Several administration routes, including external controlled subcutaneous pumps [6],
transdermal iontophoretic delivery [7] and intranasal sprays [8], have been investigated
for GnRH release stimulation. However, the electroporation route was only investigated
at room temperature [6]. The transdermal iontophoretic route may be burdensome for the
patient [7]. Intranasal administration was tested in several volunteers, but raised estrogen
levels and breakthrough bleeding were reported [8].
In considering an implantable, rhythmic delivery system for GnRH, it is helpful to
examine studies that addressed the pulsatile delivery of other drugs. Magnetic
nanohydrogels were designed to respond to pulsed magnetic fields [9]. Ca-alginate gel
beads were prepared for pulsatile dextran release. The use of various bead diameters
produced different time intervals for dextran release [10]. However, only a limited
number of pulses are possible with this formulation. Multi-layered capsules with
biodegradable layers between drug layers were proposed, but could not be realized due to
manufacturing difficulty [11].
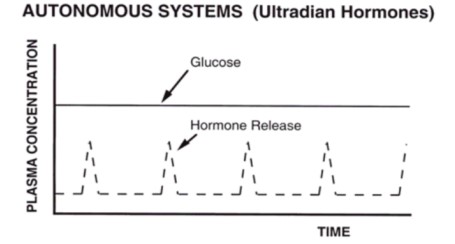
We propose to construct a hormone release system, which can produce pulsatile drug
delivery in response to endogenous stimuli, such as plasma glucose (Figure 1.1). It is
noteworthy, that this system will generate rhythmic pulses of GnRH release, even though
the blood glucose concentration remains constant. This is in contrast to system that
deliver pulses of insulin in response to changes in blood glucose level [12].
So as to release the high potency and low dose GnRH, a previous hydrogel-enzyme based
system (Figure 1.2) was achieved [13-16]. Previous publication demonstrated that under
proper conditions, the system exhibits rhythmic pulsed GnRH release with internal pH.
constant. [13]
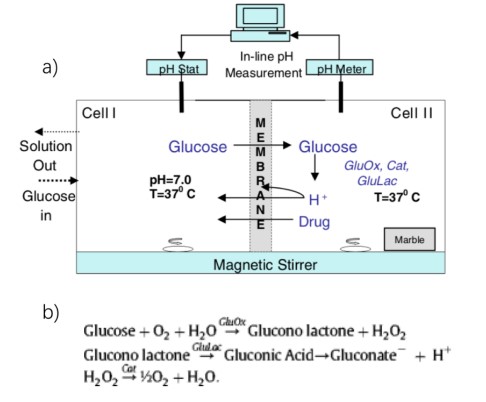
a glucose driven chemical pH oscillator. b) Enzymatic metabolism of glucose molecules
in Cell II. [14]
Figure 1.2 a) is a schematic diagram of a benchtop prototype for a drug delivery system
that is capable of releasing drug in a rhythmic, pulsatile manner. Cell I represents the
circulation, which provides a steady input of glucose and allows “waste removal” from
the system. Cell I is well stirred and maintained at 37 °C by a water jacketed beaker, and
is also pH-controlled at 7.0. Cell I is constantly fed with an aqueous glucose solution of a
specified concentration, prepared at pH 7.0, with an ionic strength of 50 mM by addition
of NaCl. Cell II also contains 50 mM NaCl aqueous solution along with enzymes glucose
oxidase (GluOx), gluconolactonase (GluLac), and catalase (Cat). Like Cell I, Cell II is
well stirred and maintained at 37 °C. Finally, Cell II contains a piece of marble (CaCO3).
Cells I and II are separated by a hydrogel consisting of poly(N-isopropylacrylamide-comethacylic acid) (NIPA/MAA), crosslinked with methylenebisacrylamide (BIS). As
discussed in Chapter 3, this membrane is charged, swollen and permeable to glucose and
to drug, when Cell II is at high pH, but is uncharged, collapsed and impermeable to
glucose and drug with Cell II is at low pH. Cell II plus the membrane together form the
prototype drug delivery system.
The proposed mechanism for the control of drug release is explained as follow. For this
pulsatile, cyclic process, it can be initially assumed that the membrane is in the charged,
swollen state. As such, glucose from Cell I will permeate the membrane into Cell II,
where it is rapidly reacted with oxygen, due to enzyme catalysis (reaction summarized in
Fig. 1.2 b). Hydrogen ion (H+
) and gluconate anion are products that cause a lowering of
pH in Cell II. The hydrogen ion then diffuses into the membrane, and neutralizes the
negatively charged, pendant carboxylate groups on the methacrylate polymer.
Eliminating the electrostatic repulsion causes the hydrogel membrane to collapse. In the
collapsed state, the membrane blocks glucose entry, and production of H+ is attenuated.
Eventually, the hydrogen ions in the membrane diffuse into Cell I and the hydrogel ion in
Cell II is neutralized by the marble, causing pH in Cell II to rise. In a time dependent
manner, determined by the properties of the membrane and cell conditions, the pH in Cell
II rises enough for the membrane to revert back to its swollen state, and glucose can reenter Cell II, restarting the cycle. As the membrane goes through cycles of swelling and
collapse, it alternatively permits and blocks drug release, giving rise to rhythmic,
pulsatile release behavior.
The previous description is ideal, insofar as it assumes that the membrane is either
permeable to glucose or is completely impermeable. In fact, even in the collapsed state,
the membrane exhibits a small but nonzero permeability to glucose. Hence, the
oscillatory behavior will not occur for all glucose concentrations in Cell I. Specifically, if
the glucose concentration is too low, then the rate of H+ generation in Cell II will be too
slow to cause sufficient neutralization for membrane collapse, and the membrane will
remain in a swollen state. On the other hand, if glucose concentration in Cell I is too
high, then there will be sufficient flux of glucose into Cell II to allow continuous
production of H+ and permanent collapse of the membrane. Thus, oscillations will only
occur in response to a range of glucose concentrations in Cell I.
Early experiments showed that this system, in the absence of marble in Cell II, slowly
evolved to a steady state of intermediate pH in Cell II and intermediate permeability to
glucose. When marble was added and glucose concentration in Cell I was increased, pH
oscillations in Cell II were observed, as was rhythmic pulsed release of drug. Marble has
the effect of consuming H+ by the reaction 2H+
+CaCO3→Ca2++CO2+H2O. This
consumption amounts to increased “clearance” of H+ from Cell II, and a higher “rate
constant” for a change of H+ concentration, i.e. pH in Cell II can change more rapidly.
Such rapid pH changes appear to be necessary to prevent the membrane from relaxing
into a state of intermediate permeability.
In this thesis, the goal was to convert the in vitro prototype, pulsatile device into a system
that can be practically implanted and provide pulsatile drug delivery. There are three key
aspects that were addressed. First, we sought to replace the commercial pH-stat that we
used, which was purchased commercially and is quite expensive, with a home-built
device. Second, a nanoclay crosslinker was evaluated as a replacement for the organic
BIS crosslinker in the hydrogel membrane to achieve improved membrane durability.
Third, the ratio of the membrane surface to the volume of Cell II was increased in an
effort to eliminate the need for marble, which would provide an alternative means to
increase the “clearance” of H+
, and hence the rate constant for change in pH in Cell II.
Additionally, we wish to optimize glucose-driven oscillation concentration at body
glucose level by reducing the membrane thickness to enhance glucose permeability.
1.3 Overview of the Thesis
Chapter 1, this chapter, presents a brief review of the medical problem of interest, several
pharmaceutical approaches to date and a brief review of a rhythmic delivery system
prototype which we hope to improve. Chapter 2 provides the working principle of a
customized pH stat as well as associated experiments used in its evaluation. Chapter 3
contains the characterization of the primary material, pH-sensitive hydrogel, for the
delivery system, with an emphasis on the properties related to device performance. The
integrated, new prototype delivery system is described in Chapter 4, along with evidence
addressing the performance of the system. Optimization methods and future direction is
summarized in Chapter 5.
Source: Nanoclay Crosslinked pH-Sensitive Hydrogel for Rhythmic Hormone Delivery

